Save time and money with our fully customizable Quality Manual and Procedures Package. Our processes are well-organized and carefully designed to work together to lead your organization to continuous improvement.
- Documents in Microsoft Word or Excel for easy customization.
- Together, the docs include the content required to address each requirement of the IATF 16949:2016 Standard
- Very clear instructions in obvious Blue Text to show you where to customize your Manual and Procedures. Treat the text in blue as “revisions” or information that is specific to your company.
- Docs are all numbered for an effective control of documented information and are integrated to work together in a seamless system.
- Related documents are referenced for effective record keeping.
- Intuitive architecture for easy Document Control. We’ve numbered QMS documents to correspond with the sections of the 16949 Standard. Registrars LOVE this feature!
- This is a downloadable product.
As you implement your IATF 16949 system you will also need checklists and training. You can save time and money by purchasing our Certification Packages!
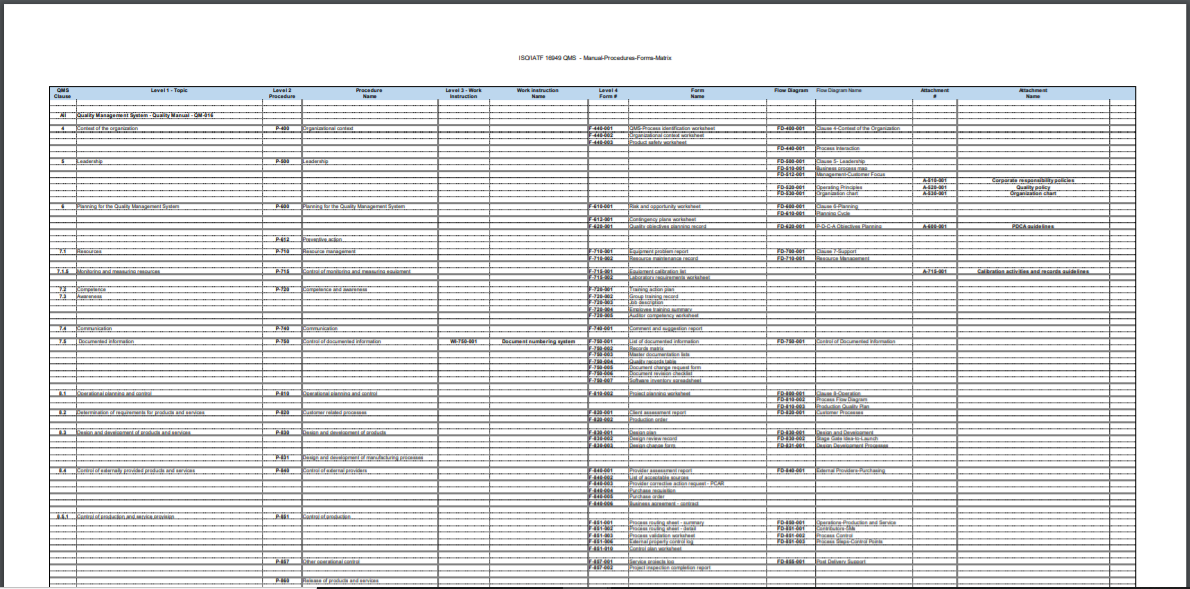
The documentation package for the management system will contain:
- (1) condensed Manual to introduce the documented information required for ISO 16949:2016.
- (28) procedures outlining how you will control each requirement of the standard
- (1) work instruction outlining document numbering system
- (63) forms to record the data required by the procedure
- (5) attachments
- (31) flow diagrams
- (3) blank, formatted templates to create your own additional documents
- Technical Support
*Includes 1 Free IATF 16949:2016 Online Training (30-days to complete training).
Compare Products
Which version is right for me?
- Choose IATF 16949:2016 to create a new IATF 16949 Quality Management System
- Choose TS 16949:2009 to IATF 16949:2016 QMS to modify your existing TS 16949:2009 QMS to meet the IATF 16949:2016 requirements
- Choose ISO 9001:2015 to IATF 16949:2016 QMS Upgrade to modify your existing ISO 9001:2015 to meet the IATF 16949:2016 requirements
If you plan to reconfigure your existing quality manual completely by yourself, you can use either of the Upgrade Instructions to create everything on your own. They will tell you where to make the changes but will NOT provide any procedure or form templates for the new requirements.
| IATF 16949 QMS |
TS 16949 to IATF 16949 QMS |
ISO 9001 to IATF 16949 QMS |
||
|---|---|---|---|---|
| Your Price | ||||
| IATF 16949:2016 Quality Manual | ||||
| IATF 16949:2016 Procedures | 28 | 28 | 4 | |
| IATF 16949:2016 Forms and Attachments | 68 | 68 | 6 | |
| IATF 16949:2016 Flow Charts | 31 | 31 | ||
| TS 16949:2009 to IATF 16949:2016 QMS Upgrade Instructions | ||||
| ISO 9001:2015 to IATF 16949:2016 QMS Upgrade Instructions | ||||
| * IATF 16949:2016 Online Training | ||||
| Support | ||||
| Your Price |
* Includes 1 Free IATF 16949:2016 Online Training (30-days to complete training).
Specific Contents
| Procedures |
|---|
| P-1010 Improvement |
| P-1020 Nonconformity and corrective action |
| P-400 Organizational context |
| P-500 Leadership |
| P-600 Planning for the Quality Management System |
| P-612 Preventive action |
| P-710 Resource management |
| P-715 Control of monitoring and measuring equipment |
| P-720 Competence and awareness |
| P-740 Communication |
| P-750 Control of documented information |
| P-810 Operational planning and control |
| P-820 Customer related processes |
| P-830 Design and development of products |
| P-831 Design and development of manufacturing processes |
| P-840 Control of external providers |
| P-851 Control of production |
| P-852 Identification and traceability |
| P-854 Preservation |
| P-857 Other operational control |
| P-860 Release of products and services |
| P-870 Control of nonconforming outputs |
| P-910 Monitoring, measurement, analysis and evaluation |
| P-911 Statistical techniques |
| P-912 Customer satisfaction |
| P-913 Root cause analysis |
| P-920 Internal audit |
| P-930 Management review |
| Forms |
| F-1010-001 Data analysis worksheet |
| F-1020-001 Corrective action request – CAR |
| F-440-001 QMS-Process identification worksheet |
| F-440-002 Organizational context worksheet |
| F-440-003 Product safety worksheet |
| F-610-001 Risk and opportunity worksheet |
| F-612-001 Contingency plans worksheet |
| F-620-001 Quality objectives planning record |
| F-710-001 Equipment problem report |
| F-710-002 Resource maintenance record |
| F-715-001 Equipment calibration list |
| F-715-001 Laboratory requirements worksheet |
| F-720-001 Training action plan |
| F-720-002 Group training record |
| F-720-003 Job description |
| F-720-004 Employee training summary |
| F-720-005 Auditor competency worksheet |
| F-740-001 Comment and suggestion report |
| F-750-001 List of documented information |
| F-750-002 Records matrix |
| F-750-003 Master documentation lists |
| F-750-004 Quality records table |
| F-750-005 Document change request form |
| F-750-006 Document revision checklist |
| F-750-007 Software inventory spreadsheet |
| F-810-002 Project planning worksheet |
| F-820-001 Client assessment report |
| F-820-002 Production order |
| F-830-001 Design plan |
| F-830-002 Design review record |
| F-830-003 Design change form |
| F-840-001 Provider assessment report |
| F-840-002 List of acceptable sources |
| F-840-003 Provider corrective action request – PCAR |
| F-840-004 Purchase requisition |
| F-840-005 Purchase order |
| F-840-006 Business agreement – contract |
| F-851-001 Process routing sheet – summary |
| F-851-002 Process routing sheet – summary |
| F-851-003 Process validation worksheet |
| F-851-006 External property control log |
| F-851-010 Control plan worksheet |
| F-852-001 Identification tag |
| F-852-002 Traceability serial number log |
| F-852-003 Traceability label |
| F-854-001 Storage inspection report |
| F-854-002 Packing slip / Invoice |
| F-857-001 Service projects log |
| F-857-002 Project inspection completion report |
| F-870-001 Nonconformance report – NCR |
| F-910-001 Production-Monitoring, measuring, and analysis table |
| F-910-002 QMS-Monitoring, measuring, and analysis table |
| F-910-004 Inspection report |
| F-911-001 Frequency distribution report |
| F-912-001 Customer survey and analysis |
| F-913-001 Root cause analysis action plan |
| F-920-001 Applicable procedure by work area |
| F-920-002 Internal audit checklist |
| F-920-003 Audit plan |
| F-920-004 Audit report |
| F-930-001 Management review agenda |
| F-930-002 Management review output report |
| Attachments & Work Instructions |
| A-510-001 Corporate responsibility policies |
| A-520-001 Quality policy |
| A-530-001 Organization chart |
| A-600-001 PDCA guidelines |
| A-715-001 Calibration activities and records guidelines |
| WI-750-001 Document numbering system |
| Flow Charts |
| FD-440-001 Process interaction flow diagram |
| FD-510-001 Business process map |
| FD-810-002 Process flow diagram |
| FD-850-001 Operations-Production and Service |